What is Sodium Hypochlorite?

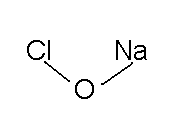
Sodium hypochlorite is a disinfectant or bleaching agent often used for the household cleaning of everyday items. It's scientific name is sodium hypochlorite. It's common name is bleach. It is composed of a sodium cation (Na+) and a hypochlorite anion (CLO-).
Compound
When was it discovered?
In 1785, a Frenchman named Berthollet developed liquid bleaching agents based on sodium hypochlorite. He then promoted his product to the Javel company whom introduced this product and named it the "liquid du Javel." It's first and primary use was to bleach cotton. This solution soon became extremely popular in France due to it's ability to remove stain from cotton clothing. It was so popular because cotton was the material of choice in all clothes at the time. In France, sodium hypochlorite or bleach is still known as the "eau du Javel" or the water of Javel.
Compound
- NaCLO
When was it discovered?
In 1785, a Frenchman named Berthollet developed liquid bleaching agents based on sodium hypochlorite. He then promoted his product to the Javel company whom introduced this product and named it the "liquid du Javel." It's first and primary use was to bleach cotton. This solution soon became extremely popular in France due to it's ability to remove stain from cotton clothing. It was so popular because cotton was the material of choice in all clothes at the time. In France, sodium hypochlorite or bleach is still known as the "eau du Javel" or the water of Javel.
Molecular Structure |
|
Home and Industry

Sodium hypochlorite is used heavily industrially and at home. Industrially, sodium hypochlorite is used as a water purifier for large scale water filtration companies. It is used industrially by janitors and cleaning staff across all fields. It is the most heavily used chemical by cleaning staff due to it's quick and efficient ability to remove stains and dirt. Additionally, it is used by some dentistry offices to remove dental stains which are caused by fluorosis. Moreover, it is also used to remove mold stains. At home, it is used to clean toilets, sinks, crockery (mostly from tea stains), and used in laundry detergents. Bleach or sodium hypochlorite is used daily at home and in industrial work. Sodium hypochlorite is also used as a disinfectant in health care buildings.
Dilution
Other Uses
Dilution
- 2% household bleach is used in the cleaning of counter tops and surfaces before the brewing of beer
- Food surfaces are allowed to be cleaned with bleach as long as they are rinsed and washed adequately
- Strong chlorine solution containing bleach is used to clean areas contaminated with bodily fluids
Other Uses
- Treat dilute cyanide waste water
- Used to kill unwanted organisms
- 50% diluted bleach is used to clean equipment that has come into contact with nerve agents
- Used to treat and reduce eczema